What are the catalysts used in the synthesis of the compound with CAS 78 - 63 - 7?
As a trusted supplier of the compound with CAS 78 - 63 - 7, I've received numerous inquiries about the catalysts involved in its synthesis. CAS 78 - 63 - 7 corresponds to 2 - Methyl - 2 - butene, an important organic compound with a wide range of applications in the chemical industry, including as an intermediate in the synthesis of various chemicals and polymers.
1. Acid - Based Catalysts
One of the most common types of catalysts used in the synthesis of 2 - Methyl - 2 - butene is acid - based catalysts. These catalysts work by protonating the reactants, which facilitates the formation of carbocations and subsequent elimination reactions.
Sulfuric acid (H₂SO₄) is a well - known acid catalyst. In the synthesis of 2 - Methyl - 2 - butene, it can be used to catalyze the dehydration of 2 - Methyl - 2 - butanol. The reaction mechanism involves the protonation of the hydroxyl group of 2 - Methyl - 2 - butanol, followed by the departure of a water molecule to form a carbocation. Then, a base abstracts a proton from an adjacent carbon atom, resulting in the formation of the double bond in 2 - Methyl - 2 - butene.
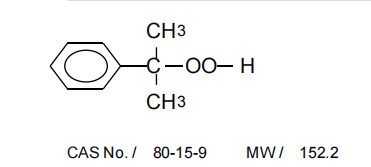

Phosphoric acid (H₃PO₄) is another acid catalyst that can be employed. It is often preferred over sulfuric acid in some cases because it is less corrosive and can provide a more controlled reaction environment. The use of phosphoric acid also leads to fewer side reactions, thus increasing the selectivity of the synthesis process.
2. Zeolite Catalysts
Zeolites are microporous, aluminosilicate minerals that have a unique crystal structure with well - defined pores and channels. They are widely used as catalysts in the chemical industry due to their high surface area, shape - selectivity, and acid - base properties.
In the synthesis of 2 - Methyl - 2 - butene, zeolite catalysts can be used to promote the isomerization and cracking reactions. For example, ZSM - 5 is a commonly used zeolite catalyst. It has a three - dimensional pore structure that can selectively adsorb and react with specific molecules. In the synthesis of 2 - Methyl - 2 - butene, ZSM - 5 can catalyze the isomerization of other butene isomers to 2 - Methyl - 2 - butene. The shape - selectivity of ZSM - 5 allows it to favor the formation of the desired product while suppressing the formation of unwanted by - products.
3. Metal - Based Catalysts
Metal - based catalysts also play an important role in the synthesis of 2 - Methyl - 2 - butene. Transition metals such as palladium (Pd), platinum (Pt), and nickel (Ni) can be used as catalysts in hydrogenation, dehydrogenation, and isomerization reactions.
Palladium catalysts are often used in hydrogenation reactions to selectively hydrogenate triple bonds or partial hydrogenation of double bonds. In the synthesis of 2 - Methyl - 2 - butene, they can be used in the hydrogenation of alkynes or in the adjustment of the degree of unsaturation of other related compounds.
Nickel catalysts are commonly used in the dehydrogenation of alkanes to form alkenes. In the case of 2 - Methyl - 2 - butene synthesis, nickel - based catalysts can be used to dehydrogenate 2 - Methylbutane to form 2 - Methyl - 2 - butene. The reaction occurs on the surface of the nickel catalyst, where the alkane molecule adsorbs, loses hydrogen atoms, and forms a double bond.
4. Peroxide - Based Catalysts
Peroxide - based compounds can also act as catalysts in some reactions related to the synthesis of 2 - Methyl - 2 - butene. For example, CHP90 (Cumene Hydroperoxide) can be used in oxidation and radical - based reactions. In some cases, it can be used to initiate radical reactions that lead to the formation of carbon - carbon double bonds.
TBHP | CAS 75 - 91 - 2 | Tert - butyl Hydroperoxide is another peroxide - based catalyst. It is a strong oxidizing agent and can be used in reactions where oxidation and activation of specific functional groups are required. For example, it can be used in the oxidation of certain alcohols or in the formation of radicals that can participate in the synthesis of 2 - Methyl - 2 - butene.
Di - Lauroyl Peroxide is a peroxide that can be used as a free - radical initiator. It decomposes to form free radicals, which can initiate polymerization or other radical - based reactions. In the context of 2 - Methyl - 2 - butene synthesis, it can be used to initiate reactions that involve the formation of carbon - carbon bonds or the isomerization of related compounds.
5. Influence of Catalysts on the Synthesis Process
The choice of catalyst has a significant impact on the efficiency, selectivity, and yield of the synthesis of 2 - Methyl - 2 - butene. A good catalyst can lower the activation energy of the reaction, thus increasing the reaction rate. For example, acid catalysts can protonate reactants and facilitate the formation of key reaction intermediates, which speeds up the dehydration or isomerization reactions.
Selectivity is also crucial in the synthesis process. Different catalysts have different selectivities towards the formation of 2 - Methyl - 2 - butene. Zeolite catalysts, with their shape - selectivity, can preferentially form the desired product while minimizing the formation of side products. This is important for reducing the cost of purification and increasing the overall quality of the final product.
The yield of the synthesis is affected by the activity and stability of the catalyst. A highly active catalyst can convert more reactants into products, while a stable catalyst can maintain its activity over a long period of time. Metal - based catalysts, for example, need to be carefully designed and prepared to ensure high activity and stability under reaction conditions.
6. Conclusion and Invitation to Purchase
In conclusion, the synthesis of the compound with CAS 78 - 63 - 7 (2 - Methyl - 2 - butene) involves a variety of catalysts, including acid - based catalysts, zeolite catalysts, metal - based catalysts, and peroxide - based catalysts. Each type of catalyst has its own unique properties and functions, and the choice of catalyst depends on the specific reaction conditions, reactants, and desired product characteristics.
As a supplier of CAS 78 - 63 - 7, we are committed to providing high - quality products and technical support. If you are interested in purchasing 2 - Methyl - 2 - butene or have any questions about its synthesis or application, please feel free to contact us for further discussions and negotiations. We look forward to establishing a long - term and mutually beneficial cooperation with you.
References
- Smith, J. H. (2015). Catalysis in Organic Synthesis. Wiley - VCH.
- Thomas, J. M., & Thomas, W. J. (2017). Principles and Practice of Heterogeneous Catalysis. Wiley.
- Ertl, G., Knözinger, H., & Weitkamp, J. (2008). Handbook of Heterogeneous Catalysis. Wiley - VCH.




