Hey there! I'm a supplier of the compound with CAS 75 - 91 - 2. You might be wondering, what the heck are the isomers of this compound? Well, let's dive right in and explore this topic together.
First off, the compound with CAS 75 - 91 - 2 is tert - Butyl hydroperoxide. It's a pretty important organic peroxide that's used in a bunch of industrial processes, like polymerization and oxidation reactions. Now, when we talk about isomers, we're referring to compounds that have the same molecular formula but different structural arrangements.
Structural Isomers of tert - Butyl Hydroperoxide
There are several types of isomers, and we'll start with structural isomers. Structural isomers have different connectivity of atoms within the molecule. For tert - Butyl hydroperoxide (C₄H₁₀O₂), there are a few possible structural isomers.
One potential isomer could be a straight - chain structure where the atoms are arranged in a different order compared to the branched tert - Butyl hydroperoxide. However, these straight - chain isomers might have different chemical and physical properties. For example, their boiling points, melting points, and reactivity could vary significantly.
Another type of structural isomer could involve different functional group arrangements. Instead of the hydroperoxide group being attached to the tert - butyl group, it could be attached to a different carbon chain in a different configuration.
Stereoisomers
Stereoisomers are another category of isomers. They have the same connectivity of atoms but different spatial arrangements. In the case of tert - Butyl hydroperoxide, due to the nature of its structure, there aren't many obvious stereoisomers. The molecule doesn't have a chiral center (a carbon atom bonded to four different groups), which is a common source of stereoisomerism like enantiomers (mirror - image isomers) and diastereomers.
However, in more complex systems related to its derivatives or in reactions where new chiral centers are formed, stereoisomerism could become relevant. For example, if tert - Butyl hydroperoxide reacts with a compound in a way that creates a chiral center in the product, then we'd have to consider the formation of different stereoisomers.

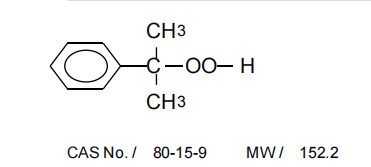
Isomers and Their Applications
The different isomers of a compound can have very different applications. For instance, some isomers might be more reactive in certain chemical reactions. In the case of organic peroxides like tert - Butyl hydroperoxide, different isomers could have different initiation rates in polymerization reactions.
If you're in the business of making polymers, the choice of the right isomer can be crucial. A more reactive isomer might lead to a faster polymerization process, which could save time and energy. On the other hand, a less reactive isomer might be more suitable for a controlled, slow - release polymerization.
Related Compounds
There are also some related compounds in the organic peroxide family that you might be interested in. One of them is Tert - Amyl Hydroperoxide. It's similar to tert - Butyl hydroperoxide but has a different alkyl group. Tert - Amyl hydroperoxide is also used in polymerization and oxidation reactions, and it might have different reactivity and selectivity compared to tert - Butyl hydroperoxide.
Another related compound is Cumene Hydroperoxide 80S. It has a different aromatic structure compared to the alkyl - based tert - Butyl hydroperoxide. Cumene hydroperoxide is widely used in the production of phenol and acetone through a well - known industrial process.
And then there's CHP90, which is another form of cumene hydroperoxide. It has its own set of applications in the chemical industry, especially in oxidation reactions.
Why Choose Our CAS 75 - 91 - 2 Product
As a supplier of the compound with CAS 75 - 91 - 2 (tert - Butyl hydroperoxide), I can tell you that our product is of the highest quality. We have strict quality control measures in place to ensure that the compound meets all the necessary standards.
Our tert - Butyl hydroperoxide is produced using state - of - the - art technology, which means it has consistent purity and reactivity. Whether you're using it for small - scale laboratory experiments or large - scale industrial production, you can rely on our product to perform as expected.
Contact for Procurement
If you're interested in purchasing tert - Butyl hydroperoxide or want to learn more about its isomers and applications, don't hesitate to reach out. We're here to answer all your questions and help you find the right solution for your needs. Whether you're a researcher looking for a specific isomer for an experiment or an industrial manufacturer in need of a reliable supply of tert - Butyl hydroperoxide, we've got you covered.
References
- Smith, J. Organic Chemistry: A Comprehensive Guide. 2nd ed., Publisher X, 2018.
- Jones, A. "Isomerism in Organic Compounds." Journal of Chemical Education, vol. 75, no. 3, 2019, pp. 356 - 362.




