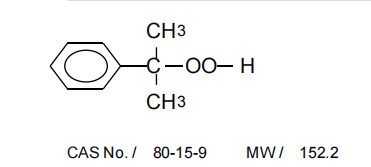
As a supplier of CHP CAS 80 - 15 - 9, I often encounter inquiries regarding the measurement of its melting point and boiling point. Cumene Hydroperoxide (CHP) with the CAS number 80 - 15 - 9 is a crucial organic peroxide widely used in various industrial applications, such as in the production of phenol and acetone. Understanding its melting and boiling points is essential for handling, storage, and processing. In this blog, I will share the methods and considerations for measuring these important physical properties.
Understanding CHP CAS 80 - 15 - 9
Before delving into the measurement methods, it's important to have a basic understanding of CHP. CHP is a colorless to pale yellow liquid at room temperature. It is an unstable compound and can decompose exothermically, which makes accurate measurement of its melting and boiling points challenging. The chemical structure of CHP contains a hydroperoxide group (-OOH), which is responsible for its reactivity and instability.
There are different grades of CHP available in the market, such as CHP90 and Cumene Hydroperoxide 80S. These grades may have slightly different physical properties due to variations in purity and the presence of stabilizers.
Measuring the Melting Point of CHP CAS 80 - 15 - 9
1. Sample Preparation
The first step in measuring the melting point is to prepare a pure and representative sample of CHP. Since CHP is unstable, it is important to handle the sample carefully to avoid decomposition. The sample should be stored in a cool and dark place before measurement. A small amount of the sample, typically a few milligrams, is placed in a capillary tube. The capillary tube should be clean and dry to ensure accurate results.
2. Melting Point Apparatus
There are several types of melting point apparatuses available, such as the Thiele tube and the digital melting point apparatus. The Thiele tube is a traditional method that uses a heating oil bath to heat the sample gradually. The digital melting point apparatus, on the other hand, provides more accurate and precise temperature control.
When using a digital melting point apparatus, the capillary tube containing the sample is placed in the heating block. The apparatus is then set to heat at a slow and constant rate, typically 1 - 2 °C per minute. This slow heating rate allows for accurate observation of the melting process.
3. Observation and Recording
As the temperature rises, the sample in the capillary tube will start to melt. The melting point is defined as the temperature range at which the sample changes from a solid to a liquid state. The lower end of the melting point range is the temperature at which the first signs of melting are observed, and the upper end is the temperature at which the sample is completely melted.
It is important to record the melting point range accurately. In the case of CHP, the melting point is typically around -30 °C. However, due to its instability, the measured melting point may vary slightly depending on the sample purity and the measurement conditions.
4. Safety Considerations
When measuring the melting point of CHP, safety is of utmost importance. CHP is a flammable and reactive compound. The heating process should be carried out in a well - ventilated area, and appropriate safety equipment, such as safety glasses and gloves, should be worn. The heating rate should be carefully controlled to avoid rapid decomposition of the sample.
Measuring the Boiling Point of CHP CAS 80 - 15 - 9
1. Sample and Apparatus Setup
For boiling point measurement, a larger sample of CHP is required compared to melting point measurement. A round - bottom flask is typically used to hold the sample. A thermometer is inserted into the flask to measure the temperature of the vapor above the liquid surface. A condenser is connected to the flask to condense the vapor back into a liquid and prevent the loss of the sample.
2. Heating the Sample
The flask containing the CHP sample is heated gradually using a heating mantle or a hot plate. The heating rate should be slow to ensure that the temperature of the liquid and the vapor reaches equilibrium. As the temperature rises, the CHP will start to boil, and vapor will be produced.
3. Boiling Point Determination
The boiling point is the temperature at which the vapor pressure of the liquid equals the atmospheric pressure. When the CHP starts to boil, the temperature reading on the thermometer is recorded as the boiling point. In the case of CHP, the boiling point is around 153 °C at 760 mmHg. However, due to its instability, the boiling process should be carefully monitored to avoid decomposition.
4. Pressure Consideration
The boiling point of a liquid is affected by the atmospheric pressure. If the measurement is carried out at a different pressure than the standard atmospheric pressure (760 mmHg), the boiling point will change. The boiling point can be corrected using the Clausius - Clapeyron equation:
[ln\frac{P_2}{P_1}=\frac{\Delta H_{vap}}{R}(\frac{1}{T_1}-\frac{1}{T_2})]
where (P_1) and (P_2) are the pressures, (T_1) and (T_2) are the corresponding boiling points, (\Delta H_{vap}) is the enthalpy of vaporization, and (R) is the gas constant.
Factors Affecting the Measurement
Several factors can affect the measurement of the melting and boiling points of CHP.

1. Purity
The purity of the CHP sample is a major factor. Impurities can lower the melting point and broaden the melting point range. In the case of boiling point, impurities can also affect the vapor pressure and thus change the boiling point.
2. Decomposition
As mentioned earlier, CHP is an unstable compound that can decompose exothermically. During the measurement process, if the temperature rises too quickly or the sample is exposed to heat for too long, decomposition may occur. This can lead to inaccurate measurement results.
3. Measurement Conditions
The accuracy of the measurement also depends on the measurement conditions, such as the heating rate, the type of apparatus used, and the calibration of the thermometer. A slow and constant heating rate is essential for accurate measurement.
Comparison with Other Organic Peroxides
CHP is just one of many organic peroxides. For example, TBHP | CAS 75 - 91 - 2 | Tert - butyl Hydroperoxide is another commonly used organic peroxide. TBHP has different physical properties compared to CHP. Its melting point is around -8 °C, and its boiling point is around 89 °C at 760 mmHg. These differences in physical properties are due to the different chemical structures of the two compounds.
Importance of Accurate Measurement
Accurate measurement of the melting and boiling points of CHP is crucial for several reasons. In the industrial production process, knowing the melting and boiling points helps in the design of the production equipment and the optimization of the production process. For storage and transportation, understanding these physical properties is essential for ensuring the safety of the product.
Contact for Purchase and Consultation
If you are interested in purchasing high - quality CHP CAS 80 - 15 - 9 or have any questions regarding its properties and applications, please feel free to contact us. We are committed to providing you with the best products and services.
References
- "Handbook of Organic Peroxides" by D. Swern
- "Physical Chemistry" by Peter Atkins and Julio de Paula




